Fluminorex Stats & Data
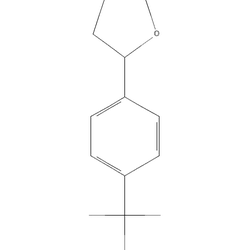
NC1=NCC(O1)c1ccc(cc1)C(F)(F)FNMGYDYBWRZHLHR-UHFFFAOYSA-NEffect Profile
CuratedStrong anxiety/jitters with moderate stimulation and focus, mild euphoria
Tolerance & Pharmacokinetics
drugs.wikiTolerance Decay
Patterns inferred from stimulant class use (amphetamine/aminorex analogues). Expect rapid tolerance with consecutive days, partial decay over 3–7 days, and near‑baseline after ~10–14 days. Evidence quality: anecdotal/low.
Cross-Tolerances
Harm Reduction
drugs.wikiFluminorex is the para‑trifluoromethyl analogue of aminorex; identity and structure are confirmed in PubChem, but there are no modern human pharmacokinetic data or clinical trials for this compound. Aminorex-class agents (including aminorex itself) have been causally associated with pulmonary arterial hypertension (PAH); mechanistically, aminorex and several anorectics act as serotonin transporter substrates, a property implicated in drug‑induced PAH, so long, repeated, or high‑dose exposure may carry non‑trivial PAH risk even if immediate subjective effects seem benign. Because of this risk, avoid daily or continuous use, set strict cycle limits, and discontinue permanently if unexplained exertional dyspnea, chest pain, syncope, or lower‑extremity edema appear; these warrant medical evaluation for PAH. The aminorex class also releases catecholamines (DAT/NET), so hyperthermia, tachycardia, and hypertension are core dose‑dependent hazards; avoid co‑use with other stimulants and manage environment (cooling, rest breaks) to reduce heat burden. Interaction risk is heightened with monoamine oxidase inhibitors (contraindicated) and with serotonergic agents (SSRIs/SNRIs/releasers/triptans) due to the class’s measurable SERT activity; combinations can precipitate hypertensive or serotonin toxicity even at moderate doses. Dosing guidance here is intentionally conservative and derived from 4‑methylaminorex user summaries (10–25 mg typical) rather than fluminorex trials; begin with threshold dosing and allow full onset before considering any increase, especially with insufflation/vaporisation where impulsive redosing risk is high. Market unfamiliarity means mislabeling is plausible; multi‑reagent kits are only presumptive and can miss adulterants — laboratory analysis (GC‑MS/LC‑MS) through a drug checking service is the most reliable way to confirm identity and detect mixtures. If using despite the above, prioritize oral dosing, spacing sessions by at least 10–14 days, maintain hydration with electrolytes while avoiding over‑hydration, and plan for insomnia with a stimulant‑free sleep window; avoid sedative ‘comedown’ cocktails (e.g., high‑dose alcohol, GHB, benzodiazepines) which carry their own risks.
References
Drugs.wiki References
- PubChem: Fluminorex (CID 24100) — identity and structure
- DrugBank article: aminorex/fenfluramine/chlorphentermine are SERT substrates; PAH implication (Circulation 1999)
- NCBI MedGen: Drug- and toxin-induced pulmonary arterial hypertension (mentions aminorex)
- EU Drugs Agency (EMCDDA/EUDA) Joint report — monoamine release potencies for aminorex/4‑MAR/4,4′‑DMAR
- Erowid 4‑Methylaminorex Basics — typical dose and duration used as proxy
- Drug checking services — methods and limitations (GC‑MS/LC‑MS)
- DrugBank: Aminorex monograph (background; amphetamine-like anorectic, PAH warning)