Arimistane Stats & Data
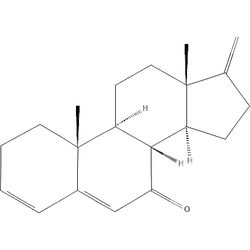
O=C1CCC2C3C(=O)C=C4C=CCCC4(C)C3CCC12CVHDOTNMSJDQVEE-YJVBKCKTSA-NEffect Profile
CuratedMild sedation
Tolerance & Pharmacokinetics
drugs.wikiTolerance Decay
No formal tolerance data. Timings reflect community patterns (weeks of continuous daily use leading to reduced responsiveness and increased side effects), not receptor-level tolerance. Interpret as practical guidance rather than a pharmacologic model.
Cross-Tolerances
Harm Reduction
drugs.wikiMechanism: Androst-7-one/androstadien-7-one derivatives inhibit aromatase competitively in human placental microsomes (not suicidally), so estrogen suppression is dose- and exposure-dependent; plan dosing assuming a competitive inhibitor rather than an irreversible binder. Labs: For any non-trivial use, obtain baseline lipids and liver enzymes and recheck periodically; estradiol monitoring should use a sensitive assay when possible to detect over-suppression. Bone and joints: Class-wide AI use is associated with arthralgia and lower bone mineral density over time; avoid extended continuous use and consider bone health if cycles are repeated. Potency variability: Over-the-counter products vary in identity and dose; start conservatively (even ≤5–12.5 mg) and titrate only with symptoms and lab feedback to avoid an E2 crash. Cycle length: Limit to the minimal effective duration; long daily use increases risk of fatigue, mood changes, libido loss, and joint pain reported by users. ROA caveats: Sublingual and transdermal routes are user-invented with unknown bioavailability; prefer oral unless you have a specific reason and can monitor. Stopping: After discontinuation, expect estrogen to rebound toward baseline within days; tapering or skip-day patterns may reduce swings. Sex-specific risks: Contraindicated in pregnancy; in premenopausal women, AIs can stimulate ovaries via feedback and are generally inappropriate outside medical care. Stacking: Combining with other anti-estrogenic agents (SERMs, strong AIs, DHT derivatives, or 5α-reductase inhibitors) can push estrogen too low; add one lever at a time and verify with labs. Lipids: AIs may worsen lipid profile; combining with 17-alkylated oral steroids further strains HDL/LDL—monitor lipids and consider discontinuation if ratios deteriorate.
References
Drugs.wiki References
- PubChem – Androsta-3,5-diene-7,17-dione (CID 150910)
- J Med Chem 1994 – Androst-5-en-7-ones and androsta-3,5-dien-7-ones inhibit aromatase competitively (DrugBank article A266865)
- NCBI StatPearls – Aromatase Inhibitors: mechanism, monitoring, contraindications
- Reddit r/prohormones – Androst 3,5-dien-7,17-dione thread (reports of E2 crash at modest dosing)
- Reddit r/prohormones – Arimistane side effects with long daily use
- Reddit r/prohormones – How to take arimistane? (community dosing cautions)
- Drugs-Forum wiki – Methandrostenolone profile (illustrates 17α-alkylated orals worsen lipid profile)