Methoxetamine Stats & Data
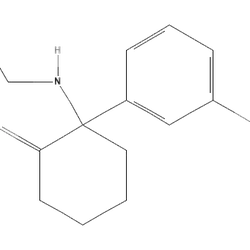
CCNC1(CCCCC1=O)c1cccc(OC)c1LPKTWLVEGBNOOX-UHFFFAOYSA-NReceptor Profile
Receptor Actions
Receptor Binding
History & Culture
Methoxetamine represents a deliberate exercise in rational drug design rather than accidental discovery. The compound was conceived by an anonymous chemist who sought to create a ketamine analogue with improved therapeutic properties. Structurally, MXE incorporates features from both ketamine and phencyclidine, sharing particular resemblance to 3-MeO-PCP while maintaining ketamine's core arylcyclohexylamine scaffold. The design philosophy behind MXE centered on two primary objectives: enhancing the antidepressant potential observed with ketamine while minimizing the urological toxicity associated with chronic ketamine use. The reasoning held that a more potent compound would require lower doses, thereby reducing the accumulation of bladder-toxic metabolites linked to ketamine-associated cystitis. The compound was also reportedly developed with potential applications for phantom limb syndrome in mind.
2010–2011
The qualitative effects of MXE were first documented on online drug forums in May 2010, with small-scale commercial availability following by September of that year. The compound's rapid proliferation drew the attention of European drug monitoring agencies; by November 2010, the European Monitoring Centre for Drugs and Drug Addiction had formally identified the substance as an emerging novel psychoactive compound. Growth in the MXE market proved substantial over the following months. By July 2011, the EMCDDA had catalogued 58 separate websites offering the compound, typically at prices ranging from 145 to 195 euros for ten grams. The substance was distributed almost exclusively through online research chemical vendors rather than traditional street-level markets, positioning it firmly within the then-emerging novel psychoactive substances economy.
2012–present
MXE developed a substantial following within psychonaut and electronic music communities during its period of availability. The dance music press documented the compound's penetration into club culture, with Mixmag reporting in January 2012 that users had coined the slang term "roflcoptr" for the substance. UK Home Office press releases subsequently referred to the drug as "mexxy," terminology that mainstream media would adopt. The compound acquired what many users describe as a cult status, valued for a distinctive quality frequently characterized as "magic"—combining pronounced euphoria, depth of introspection, and remarkable versatility across dosage ranges. Users praised its psychedelic character relative to other dissociatives and its suitability for both social settings and solitary exploration at higher doses. However, regulatory action—particularly production bans in China, which had served as the primary manufacturing source—severely disrupted global supply. The compound became progressively more difficult to obtain, and by the mid-2010s was largely considered extinct from the research chemical marketplace. Contemporary discussions of MXE among former users often carry a tone of nostalgia, with one commentator noting that people speak of the substance in terms typically reserved for a departed lover or a prize catch that somehow escaped.
Subjective Effect Notes
cognitive: The general head space of MXE is often described as particularly euphoric and clear headed in comparison to that of DXM and Ketamine.
Effect Profile
Curated + 219 ReportsStrong dissociative depth and insight with moderate mania and motor impairment
User Experiences
Duration Timeline
BluelightCommunity Effects
TripSitTolerance & Pharmacokinetics
drugs.wikiTolerance Decay
A single heavy session can produce pronounced short-term tolerance. Many users report tolerance halves over ~3–5 days and returns near-baseline after ~10–14 days; data are anecdotal and vary widely by dose pattern and ROA. Cross-tolerance within the arylcyclohexylamine class is expected. Data quality: anecdotal/community reports.
Cross-Tolerances
Experience Report Analysis
ErowidDemographics
Gender Distribution
Age Distribution
Reports Over Time
Effect Analysis
ErowidEffects aggregated from 219 experience reports (219 Erowid)
Effect Sentiment Distribution
Confidence Distribution
Positive Effects 11
Adverse Effects 12
Dose-Response Correlation
How effect frequency changes across dose levels
Insufflated
View data table
| Effect | Common (n=16) | Strong (n=18) | Heavy (n=15) |
|---|---|---|---|
| Visual Distortions | 50.0% | 66.7% | 73.3% |
| Color Enhancement | 50.0% | 66.7% | 53.3% |
| Dissociation | 37.5% | 61.1% | 60.0% |
| Confusion | 37.5% | 38.9% | 60.0% |
| Music Enhancement | 56.2% | 50.0% | 46.7% |
| Stimulation | 31.2% | 55.6% | 40.0% |
| Euphoria | 50.0% | 44.4% | 46.7% |
| Tactile Enhancement | 25.0% | 50.0% | 20.0% |
| Empathy | 18.8% | 44.4% | 46.7% |
| Focus Enhancement | 25.0% | 38.9% | 40.0% |
| Anxiety | 31.2% | 33.3% | 26.7% |
| Motor Impairment | 31.2% | 16.7% | 33.3% |
| Closed-Eye Visuals | 12.5% | 33.3% | 33.3% |
| Nausea | 12.5% | 33.3% | 0% |
| Auditory Effects | 12.5% | 33.3% | 26.7% |
Oral
View data table
| Effect | Strong (n=11) |
|---|---|
| Music Enhancement | 81.8% |
| Anxiety | 72.7% |
| Visual Distortions | 54.5% |
| Confusion | 54.5% |
| Closed-Eye Visuals | 36.4% |
| Color Enhancement | 36.4% |
| Empathy | 36.4% |
| Focus Enhancement | 27.3% |
| Headache | 27.3% |
| Dissociation | 27.3% |
| Hospital | 27.3% |
| Introspection | 18.2% |
| Open-Eye Visuals | 18.2% |
| Euphoria | 18.2% |
| Time Distortion | 18.2% |
Subjective Effect Ontology
Experience ReportsStructured effect tags extracted from 219 experience reports using a controlled vocabulary of 220+ canonical effects across 15 domains.
Auditory
Emotional
Selfhood
Visual
Dose–Effect Mapping
Experience ReportsHow reported effects shift across dose tiers, based on 219 experience reports.
| Effect | Common (n=16) | Strong (n=18) | Heavy (n=15) | |
|---|---|---|---|---|
| visual distortions | ↑ | |||
| color enhancement | → | |||
| dissociation | ↑ | |||
| confusion | ↑ | |||
| music enhancement | ↓ | |||
| stimulation | ↑ | |||
| euphoria | → | |||
| tactile enhancement | ↓ | |||
| empathy | ↑ | |||
| focus enhancement | ↑ | |||
| anxiety | → | |||
| motor impairment | → | |||
| closed-eye visuals | ↑ | |||
| nausea | — | ↑ | ||
| auditory effects | ↑ | |||
| introspection | ↓ | |||
| sedation | → | |||
| body high | — | ↑ | ||
| open-eye visuals | — | — | → | |
| jaw clenching | — | — | → |
Showing top 20 of 26 effects
| Effect | Strong (n=11) | |
|---|---|---|
| music enhancement | ||
| anxiety | ||
| visual distortions | ||
| confusion | ||
| closed-eye visuals | ||
| color enhancement | ||
| empathy | ||
| focus enhancement | ||
| headache | ||
| dissociation | ||
| hospital | ||
| introspection | ||
| open-eye visuals | ||
| euphoria | ||
| time distortion | ||
| body high | ||
| stimulation | ||
| motor impairment | ||
| sedation | ||
| auditory effects |
Showing top 20 of 21 effects
Risk Escalation
Sentiment AnalysisAverage frequency of positive vs adverse effects across dose tiers (Insufflated)
View effect breakdown
Adverse Effects
| Effect | Common (n=16) | Strong (n=18) | Heavy (n=15) | Change |
|---|---|---|---|---|
| Confusion | +60% | |||
| Anxiety | -14% | |||
| Motor Impairment | 6% | |||
| Nausea | — | +166% | ||
| Jaw Clenching | — | — | 0% | |
| Memory Suppression | — | -20% | ||
| Headache | — | — | 0% |
Positive Effects
| Effect | Common (n=16) | Strong (n=18) | Heavy (n=15) | Change |
|---|---|---|---|---|
| Color Enhancement | 6% | |||
| Music Enhancement | -16% | |||
| Stimulation | +28% | |||
| Euphoria | -6% | |||
| Tactile Enhancement | -20% | |||
| Empathy | +148% | |||
| Focus Enhancement | +60% | |||
| Introspection | -57% | |||
| Body High | — | +19% | ||
| Pain Relief | — | — | 0% | |
| Creativity Enhancement | — | — | 0% |
Dosage Distribution
Dose distribution from experience reports
Insufflated
Oral
Sublingual
Real-World Dose Distribution
62K DosesFrom 303 individual dose entries
Insufflated (n=160)
Oral (n=52)
Intramuscular (n=18)
Rectal (n=8)
Sublingual (n=25)
Common Combinations
Most co-occurring substances in experience reports
Form / Preparation
Most common forms and preparations reported
Body-Weight Dosing
Dose relative to body weight from reports with weight data
Insufflated
Oral
Sublingual
Redose Patterns
Redosing behavior across 165 reports
Legal Status
| Country | Status | Notes |
|---|---|---|
| Austria | Illegal (SMG) | Prohibited under the Suchtmittelgesetz since June 26, 2019. Possession, production, and sale are criminal offenses. |
| Brazil | Controlled (Portaria SVS/MS nº 344) | Listed as a controlled substance on February 18, 2014 alongside several other novel psychoactive substances. Possession, production, and sale are illegal. |
| Canada | Schedule I CDSA (analogue) | Health Canada declared MXE a controlled analogue of ketamine under Schedule I of the Controlled Drugs and Substances Act in January 2011. Possession, production, and sale are prohibited. |
| China | Category I Psychotropic | Controlled as a Category I psychotropic substance. It is illegal to sell, buy, import, export, or manufacture without authorization. China's ban on production significantly impacted global MXE availability. |
| Croatia | Controlled (analogue) | Reportedly controlled as an analogue of ketamine under national drug legislation. |
| Cyprus | Controlled | Listed in the national drug control law since 2012. Possession, production, and distribution are prohibited. |
| Denmark | Controlled | Covered by the Executive Order on Euphoriant Substances, making possession, production, and distribution illegal. |
| France | Stupéfiant | Added to the list of controlled stupefiant substances on August 5, 2013. Possession, production, and distribution are illegal. |
| Germany | Anlage I BtMG | Controlled under the Betäubungsmittelgesetz since July 2013. Manufacturing, importing, possessing, selling, or transferring without license is prohibited. |
| Italy | Controlled | Listed in the table of controlled drugs since 2016. Possession, production, and distribution are prohibited. |
| Japan | Narcotic | Classified as a narcotic drug effective June 26, 2016. Earlier controls from July 2012 restricted importation, production, and distribution while possession for personal use remained in a legal grey area. |
| Netherlands | List I (Opiumwet) | Added to List I of the Opium Law in July 2015. Possession, production, transport, import, export, and sale are prohibited. |
| Russia | Controlled | Prohibited since October 2011. Possession, production, and sale are illegal under Russian narcotics legislation. |
| Slovenia | Controlled | Listed as a controlled substance per Official Gazette of the Republic of Slovenia No. 62/2013. |
| Sweden | Narcotic | Controlled under the Narcotic Drugs Control Act (SFS 1992-860) and the Narcotic Drugs Control Ordinance (SFS 1994:1554). |
| Switzerland | Controlled (Verzeichnis D) | Specifically named under Verzeichnis D of the Swiss narcotics legislation. Possession, production, and sale are illegal. |
| Turkey | Controlled | Regulated under the Law on Control of Narcotics no. 2313. Unauthorized possession, production, and distribution are prohibited. |
| United Kingdom | Class B | Permanently classified in February 2013 following a Temporary Class Drug Order issued in April 2012. The Advisory Council on the Misuse of Drugs recommended scheduling after reviewing emerging evidence of harm. |
| United States | Schedule I | Federally scheduled on July 6, 2022 under the Controlled Substances Act. Prior to federal scheduling, multiple states enacted their own controls including Alabama (2015), Arizona (2014), Florida (2012), Indiana (2012), Louisiana (2013), Minnesota (2012), North Dakota (2012), Ohio (2012), Virginia (2012), and Utah. |
Harm Reduction
drugs.wiki- Variable potency and frequent misrepresentation have been reported; some user reports historically attributed to MXE were actually other dissociatives. Always test with reagents and, where possible, obtain confirmation via analytical drug checking (e.g., GC–MS/LC–MS). Volumetric dosing reduces large weighing errors. Evidence: Erowid MXE dosage note warns about misrepresented samples; Erowid experience category “What Was in That?” documents mislabeling cases; Erowid dosing articles explain liquid measurement to improve accuracy. - Acute clinical presentations can combine ketamine-like dissociation with sympathomimetic signs (tachycardia, hypertension). Low-dose benzodiazepines were used in case series management. Evidence: ED case series of analytically confirmed MXE exposures. - A distinct acute cerebellar toxidrome (ataxia, nystagmus, incoordination) has been repeatedly described after insufflation; symptoms may last beyond the main psychoactive phase but are usually reversible. Evidence: three analytically confirmed cases. - Chronic harms: animal and in vitro data indicate MXE can induce bladder inflammation/fibrosis and renal toxicity similar to ketamine cystitis; monitor for urinary frequency, urgency, dysuria, or hematuria and stop use if present. Evidence: mouse and rat studies and urothelial cell data. - Pharmacology: besides NMDA antagonism, MXE inhibits monoamine transporters with greatest potency at SERT (low micromolar), so serotonergic combinations may be more unpredictable than with ketamine. Active metabolites (O-desmethyl- and N-desethyl-MXE) retain NMDA-blocking activity and may extend/alter effects. Evidence: in vitro neuropharmacology profiles; NMDA IC50 data for MXE and metabolites. - Set/setting: high doses can cause immobility and loss of consciousness; use a sober sitter, clear physical hazards, and avoid heights/bodies of water. Evidence: harm-reduction guidance and effects summaries noting accident susceptibility. - ROA safety: avoid IV due to unknown cuts and insolubles; if choosing IM, use sterile water or saline, micron filtration, new sterile equipment, proper needle size/site, and aseptic technique to reduce abscess/infection risk. Evidence: Erowid injection basics; community harm-reduction guidance on filtration and sterile prep. - Nasal care: powders can irritate/anaesthetize mucosa; gentle isotonic saline rinse after sessions can reduce local irritation. Evidence: community HR guidance. - Driving/coordination: residual disequilibrium can last into the next day; do not drive or operate machinery until fully baseline. Evidence: TripSit duration (after-effects up to 48 h) and Erowid effects (accident susceptibility). - Legal risks: MXE is controlled in many jurisdictions (e.g., U.S. federally scheduled since July 6, 2022); penalties vary by region. Verify local law before possession. Evidence: Erowid legal status summary.
References
Data Sources
Cited References
- Advisory Council on the Misuse of Drugs (ACMD) Methoxetamine Report 2012
- Alcohol and Drug Foundation - MXE Facts
- Anodyne.wiki Entry on MXE
- Bluelight: Big & Dandy MXE Thread
- Coppola M, Mondola R - Methoxetamine: From Drug of Abuse to Rapid-Acting Antidepressant
- DrugFacts UK - MXE Duration and Harms
- Erowid: Methoxetamine Dose Chart
- European Monitoring Centre - Online Sales of New Psychoactive Substances
- Halberstadt AL, et al. - Novel Ketamine Analog Methoxetamine Produces Dissociative-Like Behavioral Effects in Rodents
- Hi-Ground Harm-Reduction Leaflet
- Hofer KE, et al. - Ketamine-Like Effects After Recreational MXE Use
- Kjellgren A, Jonsson K - Phenomenological Study of MXE Experiences
- Meyer MR, et al. - MXE Metabolism Including Isoenzyme Kinetics and Toxicological Detectability
- Morris H - Interview with a Ketamine Chemist
- Morris H, Wallach J - From PCP to MXE: Comprehensive Review of Non-Medical Use of Dissociative Drugs
- Pain Medicine - Self-Medication Case Report
- Roth BL, et al. - Detailed Pharmacological Evaluation of MXE
- ScienceDirect: Topics - Methoxetamine Duration by Route
- UNODC: New Psychoactive Substances Report
- Wood DM, et al. - Reversible Cerebellar Toxicity Case Report
- Zanda MT, et al. - MXE Pharmacokinetics and Metabolites Review
- Detailed pharmacological evaluation of MXE
- Ketamine-like effects after recreational MXE use
- Reversible cerebellar toxicity case report
- Phenomenological study of MXE experiences
- MXE pharmacokinetics and metabolites review
- MXE NMDA IC50 data
- Pain Medicine – self-medication case
Drugs.wiki References
- Erowid MXE Vault (common names)
- Erowid MXE Dose Chart
- TripSit Wiki – Methoxetamine (duration, HR, interactions)
- Erowid MXE Effects page (risks, accident susceptibility)
- PubMed – Acute toxicity with recreational MXE (ED case series)
- PubMed – MXE-associated reversible cerebellar toxicity (3 cases)
- PubMed – Rat/mouse bladder–renal toxicity of MXE
- PubMed – Mouse bladder/renal toxicity after 3 months MXE
- PubMed – Neuropharmacological characterization (SERT/NET/DAT inhibition)
- PubMed – MXE and metabolite NMDA-blockade potencies
- Hi‑Ground Methoxetamine page (HR overview)
- Hi‑Ground MXE HR leaflet (AKAs, dose ranges, risks)
- EUDA (EMCDDA) – Council Implementing Decision (harms, deaths)
- Erowid – Liquid Measurement Technique (volumetric dosing)
- Erowid – Importance of Measured Doses
- Erowid – Syringe & Needle Basics (sterile technique)
- Bluelight – Micron filter & IM preparation HR discussion
- Drugs‑Forum – Saline nasal rinse guidance after snorting
- Erowid – MXE Experience Vaults: What Was in That?
- Erowid – MXE Legal Status (US federally scheduled July 6, 2022)